Compikal's mission is to provide its medical device customers with a productive, effective, and affordable quality management system.Compikal's vision is to become the preferred partner for advanced QMS solutions.Compliance without the headache.
Say goodbye to endless QMS maintenance with our HI expertise combined with AI-enhanced approach.Less complexity. More innovation.
Compikal keeps your quality management system up-to-date
so you can focus on building innovative medical devices.
Need to watch well-structured QMS ?View our curated QMS documentation of 40+ essential SOPs and Quality Manual with ISO 13485 & QSR 820 & ISO 14971 & applicable Reporting standards.Simply push the button and apply for FREE view:
Need QMS Health Check - Up?Utilize our Compliance Radar
to pinpoint gaps & improvement opportunities.
Comprehensive assessment aligned with ISO 13485 & QSR 820 & ISO 14971 & Reporting.
Optional expansion to EU MDR, ISO 14971, IEC 62304 & more.Starting at $649
Looking for productivity?Customizable MS Word SOPs and templates pre-built for ISO 13485, QSR 820, ISO 14971, and reporting compliance for quick and easy adaptation.
Can be extended to additional standards like IEC 62304, IEC 62366, and EU MDR.Starting at $1,449
Why choose Compikal as your QMS partner ?The Perfect Blend of HI and AI .Created by MD and IVDR industry veterans who understand your challenges.Our solutions are practical focusing on Compliance, Excellence and Productivity.
Continuous support to our customers:Free 24/7 human-powered query Q&ASharing updates on industry best practicesPatient safety focus
© Untitled. All rights reserved.
QMS Quality Manual cover page
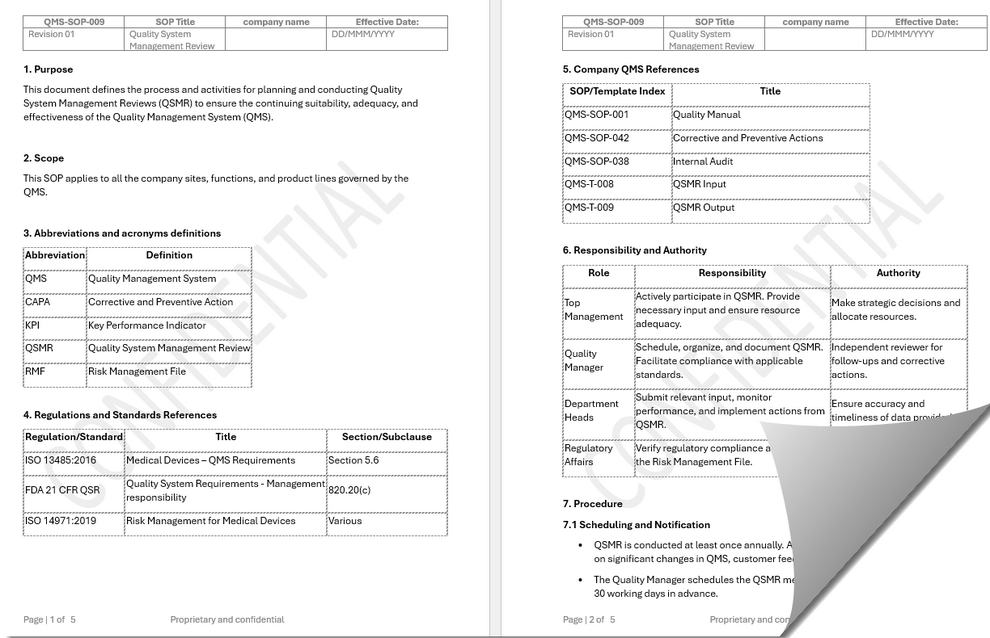
SOP QSMR front page
SOP structure
QMS Check Up report
Legal Disclaimer and Terms of Use
Compikal provides quality management and regulatory consulting services to medical device companies. While we strive to ensure the accuracy and completeness of all information, advice, and documentation provided, we make no warranties or representations regarding their fitness for any particular purpose.
Customers are solely responsible for the implementation, adaptation, and use of any information, advice, or documentation provided by Compikal. It is the customer's responsibility to ensure all quality systems and documentation comply with applicable regulations, standards, and requirements specific to their products, intended markets, and organizational needs.
The information and documentation provided by Compikal are intended as guidance only and do not constitute legal advice. Customers should consult with qualified legal counsel regarding specific regulatory requirements applicable to their medical devices.
Compikal shall not be liable for any direct, indirect, incidental, consequential, or special damages arising out of or in connection with the use of our services, documentation, or advice, regardless of whether Compikal has been advised of the possibility of such damages.
By using Compikal's services, customers acknowledge and agree to these terms.
Privacy Statement
Effective Date: March 6, 2025
At Compikal, we respect your privacy and maintain a simple approach to data protection.
Our Privacy Practices
Minimal Collection: We collect only basic contact information (name, email, company) when you request samples or contact us.
Limited Usage: Your information is used solely to respond to your inquiries and provide our services.
No Data Sharing: We do not sell, trade, or transfer your information to third parties unless required by law.
Technical Information: Our website is hosted on Carrd, which uses minimal technical resources required for basic functionality. Any third-party features (such as our chat widget) may use their own cookies or similar technologies for essential functionality only.
Security: We implement appropriate security measures to protect any information you provide.